PEPFAR

Supporting HIV-Related Laboratory Networks and Partnerships to facilitate laboratory strengthening and management activities for countries supported under the President’s Emergency Plan for AIDS Relief (PEPFAR)
What is This Project About?
This project is a unique partnership of Africa-based organisations with technical expertise, outreach to the laboratory community, and networks and political mandate to advance laboratory Medicine.
It is funded by PEPFAR to assist supported countries to further advance and sustain access to quality-assured HIV laboratory services, and bridge the gaps towards the UNAIDS 90-90-90 targets. The project assists countries, local NGOs and implementing partners to develop and adopt frameworks for the implementation of quality-assured HIV diagnostics policies and procedures in all sub-Saharan African regions.
What We Do
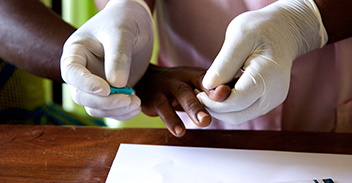
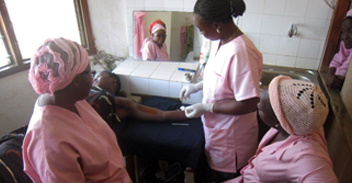
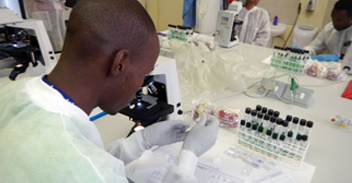

Achievements At Year One
Anticipated Outcomes



Strategic Approach
- Direct technical assistance to PEPFAR–supported countries
- Synergising partnership to reduce overlap or interventions, save on resources and create opportunities for new knowledge and innovations
- Translation of best practices into validated frameworks, policy and regulation at the national and regional level
- Dissemination of best practices and knowledge to the larger community of laboratory medicine professionals and healthcare on the continent
- Maintain a communication between Ministries of Health, partners and civil society in support of improved HIV diagnostic services and functional laboratory networks
Scope of Work







For more information
Contact the Project Lead, Dr Collins Otieno Odhiambo here