The ASLM Academy is a platform where medical laboratory professionals from Africa and across the globe can access online, face-to face training and information packages that can be used towards continuous professional development. The ASLM Academy focuses on delivering practical, problem-solving learning packages that support the advancement of knowledge and skills in laboratory medicine. It does not replace formal pre-service education, rather it assists in recognising the value of various in-service trainings for individual career development.
What We Do
Global Health Security in Africa
Finding Solutions and Emphasising the Critical Role of Laboratories:
The recent Ebola outbreak in West Africa and global COVID-19 pandemic has highlighted the importance of early disease detection and response in preventing the rise of new global health threats. ASLM is committed to supporting laboratory systems and networks that are efficient in combating global health security risks.
Updates on the Freetown, Sierra Leone, Regional Global Health Security Consultation:
- New ‘Freetown Declaration’ advocates for increased prioritisation of functional tiered laboratory networks to close gaps in global health security agenda
- View the press release from the Regional Freetown Consultation.
Early detection requires that rapid disease testing capacity reaches across countries and into rural communities where people live. Health experts in Africa are taking steps to strengthen laboratory networks in order to improve the early detection of outbreaks on the continent.
Significant progress has been made in strengthening laboratory networks since the adoption of the 2008 Maputo Declaration for Strengthening Laboratory Systems and Resolution AFR/RC58/R2 at the 58th session of the World Health Organization Regional Committee for Africa (WHO AFRO), both resulting in the scale-up of diagnostic services for HIV, tuberculosis, and malaria. Emerging threats now require even more responsive and harmonised laboratory networks, which are significantly integrated and aligned with public health institutes for effective surveillance responses.
Together with ASLM and WHO AFRO, high-level Ministry of Health officials from more than 20 countries in Africa have issued a “Freetown Declaration” calling for international and local stakeholders to bolster capacity to establish resilient tiered laboratory networks, regularly measure progress with a standardised score card, and effectively integrate these networks into disease surveillance and public health institutes.
Diagnostic Evidence Hub
The Diagnostic Evidence Hub is a knowledge platform that provides national reference laboratories, national regulatory authorities, and diagnostics stakeholders with key information from published studies on the technical performance of new in vitro diagnostic products. It seeks to improve access to publicly available technical data in order to inform decision-making and support in-country registration and adoption of new, impactful, and quality-assured diagnostic products. Click below to learn more and explore the available products.
SLIPTA
 In 2009, World Health Organization’s Regional Office for Africa (WHO AFRO) established a framework for improving the quality of public health laboratories in developing countries to achieve ISO 15189 standards. This framework, renamed in 2011 and implemented by ASLM in Africa, is the Stepwise Laboratory Quality Improvement Process Towards Accreditation (SLIPTA) programme.
In 2009, World Health Organization’s Regional Office for Africa (WHO AFRO) established a framework for improving the quality of public health laboratories in developing countries to achieve ISO 15189 standards. This framework, renamed in 2011 and implemented by ASLM in Africa, is the Stepwise Laboratory Quality Improvement Process Towards Accreditation (SLIPTA) programme.
Through standardised processes, SLIPTA measures and evaluates the progress of laboratories towards international accreditation and awards a certificate of recognition with 0-5 star ratings. SLIPTA enables laboratories to develop their quality management systems in order to produce timely, reliable and accurate laboratory results.
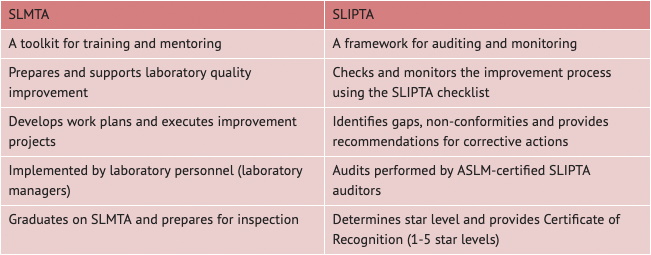
What is the difference between SLIPTA and SLMTA?
Learn MoreProportion of internationally accredited medical/clinical laboratories in Africa from 2014 and 2019
Since the launch of the SLIPTA programme and the Maputo Declaration, the number of accredited laboratories in Africa has increased from 422 in 2014 (Figure 1) to 633 in 2019 (Figure 2). As indicated in the figures, there the proportion of laboratories achieving accreditation outside South Africa is increasing, illustrating the overall advancement of quality management systems across the continent. Twenty percent of non-South African laboratories achieving ISO accreditation in 2019 have been engaged in the SLIPTA programme.
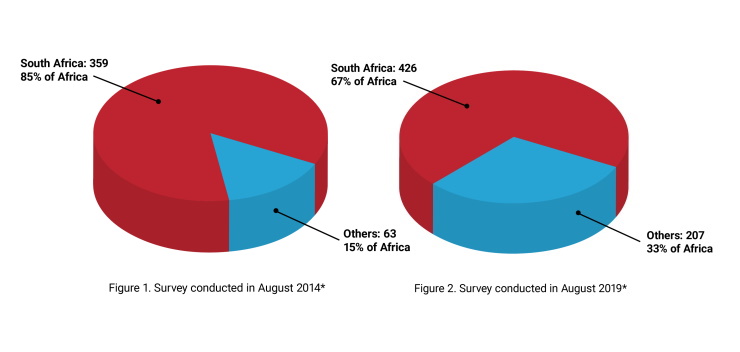 *Data collected from these accreditation bodies
*Data collected from these accreditation bodies
See the latest SLIPTA Audited Laboratories Distribution Map here.
See the WHO Guide for the Stepwise Laboratory Improvement Process Towards Accreditation in the African Region (with checklist) here.
View All SLIPTA ResourcesAffiliations

ISO TC 212 is the global ISO committee that addresses the field of Laboratory Medicine. As a liaison organization of ISO TC212, ASLM contributes to the work of the technical committee and subcommittees.

AFRAC is a cooperation of accreditation bodies, sub-regional accreditation collaborations and stakeholders whose objective is to facilitate trade and contribute to the protection of health, safety and the environment in Africa. ASLM is a stakeholder of AFRAC and contributes by facilitating capacity building in accreditation across Africa with the goal of sustaining an internationally acceptable, mutual recognition arrangement.
PEPFAR Project
The PEPFAR project is a unique partnership of Africa-based organisations with technical expertise, outreach to the laboratory community, and networks and political mandate to advance laboratory medicine.
Learn MoreAntimicrobial Resistance
Antimicrobial Resistance (AMR) kills 70,000 patients annually. ASLM works with countries and stakeholders to move the agenda towards the containment of AMR in Africa through laboratory-based surveillance and clinical testing.
Learn MoreLabCoP
The Laboratory Systems Strengthening Community of Practice (LabCoP) is a new learning network, designed to link multidisciplinary teams from member countries to exchange laboratory experiences and best practices.
Learn MoreLabMaP
ASLM’s role in the laboratory mapping program is to propose solutions for the standardised collection, storage and analysis of geo-located data on laboratory capacity, and to support the establishment of public and country portals in accordance with national requirements for data sharing.
Learn MoreIntegrated Diagnostics Consortium
The global market for diagnostics in low-resource settings does not meet the needs of the millions of people living with HIV, TB, HPV, Hepatitis and other diseases, so the Integrated Diagnostics Consortium was formed to enable the scale-up of better coordinated procurement and deployment of platform-based diagnostic technologies.
Learn MoreOutbreak Coverage
Here you will find the latest news, guidelines and tools to fight endemic and pandemic outbreaks.
Learn MorePoint-of-Care Testing
 Point-of-care (POC) technologies bring tests closer to patients in a convenient and timely manner to allow rapid care and treatment. The introduction of molecular testing has been a game changer, particularly for major diseases such as tuberculosis and pediatric HIV. Tests for these diseases traditionally entailed transporting samples from local clinics to testing centres, and patients and clinicians often waiting hours, weeks or months, to receive results needed to inform care and treatment. Today, HIV test results are usually returned to caregivers on the same day and HIV-infected infants can begin antiretroviral therapy (ART) immediately, reducing the risk of loss to follow up. However, in many settings, obstacles remain to POC testing even when POC testing is available, and must be overcome if, the full potential of POC early infant diagnosis (EID) is to be maximised.
Point-of-care (POC) technologies bring tests closer to patients in a convenient and timely manner to allow rapid care and treatment. The introduction of molecular testing has been a game changer, particularly for major diseases such as tuberculosis and pediatric HIV. Tests for these diseases traditionally entailed transporting samples from local clinics to testing centres, and patients and clinicians often waiting hours, weeks or months, to receive results needed to inform care and treatment. Today, HIV test results are usually returned to caregivers on the same day and HIV-infected infants can begin antiretroviral therapy (ART) immediately, reducing the risk of loss to follow up. However, in many settings, obstacles remain to POC testing even when POC testing is available, and must be overcome if, the full potential of POC early infant diagnosis (EID) is to be maximised.
POC Testing is critical because:
- It brings essential diagnostic testing closer to patients, therefore quicker access to results
- It helps more children receive more access to life-saving care
- It facilitates more rapid clinical decision making
- Point-of-care viral load (VL) testing can address systemic challenges and support PMTCT efforts
- It can help avert vertical transmission from pregnant or breastfeeding mothers to their children
- It facilitates more appropriate treatments and interventions

ASLM has partnered with The Clinton Health Access Initiative (CHAI) and UNICEF to accelerate adoption and uptake of new diagnostic technologies under the Unitaid-funded POC HIV Diagnostic Project, which brings essential diagnostic testing closer to patients. Together with the Elizabeth Glaser Pediatric AIDS Foundation (EGPAF), ASLM, CHAI, UNICEF and Unitaid, we have successfully supported the introduction of POC EID and VL technologies, pilot and scale up activities in 15 sub-Saharan African countries.
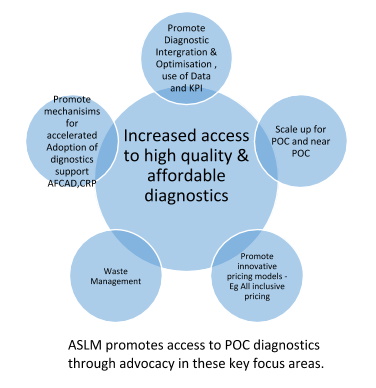 In order to accelerate POC implementation ASLM disseminates related resources, evidence and lessons learned, leading the development of technical best practices of supportive systems, and establishing a community of practice for laboratory leaders, implementers, and partners to promote knowledge sharing and south-to-south learning.
In order to accelerate POC implementation ASLM disseminates related resources, evidence and lessons learned, leading the development of technical best practices of supportive systems, and establishing a community of practice for laboratory leaders, implementers, and partners to promote knowledge sharing and south-to-south learning.
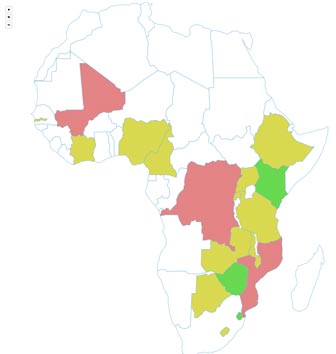 The number of types of technologies formally registered per country can been used as a proxy indicator for uptake of this relatively new market of POC NAT diagnostics. This map indicates the distribution by type and numbers of POC Nucleic Acid Testing (NAT) for HIV, EID and VL technologies being used by some countries in Africa.
The number of types of technologies formally registered per country can been used as a proxy indicator for uptake of this relatively new market of POC NAT diagnostics. This map indicates the distribution by type and numbers of POC Nucleic Acid Testing (NAT) for HIV, EID and VL technologies being used by some countries in Africa.
ASLM and our partners have amassed a collection of tools, guidelines, publications, reports, presentations and other resources related to point-of-care, viral load testing, waste management and other relevant topics, to help build your knowledge base and enhance your organisation.
Centres of Excellence
Centres of excellence strengthening laboratory medicine and improving health outcomes in Africa
ASLM’s Network of Centres of Excellence comprise a nucleus of the leading national public health laboratories on the African continent. These Centres of Excellence play a key role in strengthening laboratory medicine and improving health outcomes in Africa by becoming leaders in several important initiatives.
This ASLM laboratory network currently consist of six centres of excellence with a national and international reputation in laboratory medicine, research, policy guidance, technology assessment and implementation, and training, amongst other areas. Three of the centres are accredited to international ISO standards.
Significant opportunities exist for the ASLM network of Centres of Excellence to strengthen laboratory medicine in Africa, including:
- Improving regulatory harmonisation for diagnostics within economic zones. Opportunities exist for theC entres of Excellence to partner in efforts with other agencies to establish regional diagnostics regulatory harmonisation. Several groups, including WHO, ASLM, the London School of Hygiene and Tropical Medicine (LSHTM), and others are working on initiatives to build the appropriate political support, create assessment standards etc. The Centres of Excellence can play a key role by conducting evaluations of new technology to produce high quality data for regional regulatory review.
- Establishing a south-south fellowship programme to provide opportunities for promising young laboratory scientists to train, study, and research at centres of excellence for several months at a time. This would promote skills transfer and collaboration between the network of laboratories on the continent and help develop the next generation of laboratory medicine leaders.
- Establishment of an international network of proficiency testing (PT) providers to support SLIPTA and other quality improvement and accreditation programmes. Each Centre of Excellence would be responsible for a different range of PT services as part of a self-sustaining initiative, coordinated by a committee of the Centres of Excellence with a rotating lead.
- Fostering the establishment of the Association/Network of National Public Health Reference Laboratories (NPHL) in Africa. NPHLs play a key role in training, assuring quality and developing laboratory capacity throughout the health care system in their respective countries. Linking these laboratories in a regional network is critical to increase research capacity, quality management systems, disease detection, and sustainability of health programs throughout Africa. Moreover, the regional laboratory network will facilitate South-South cooperation and collaboration.
ASLM’s Network of Centres of Excellence: